|
3/11/2024 0 Comments Bohr model of hydrogen atom In effect, an atom can “store” energy by using it to promote an electron to a state with a higher energy and release it when the electron returns to a lower state. The great Danish physicist Niels Bohr (18851962) made immediate use of Rutherford’s planetary model of the atom. First, and perhaps most strikingly, the Schrödinger model does not produce well-defined orbits for the electron. There are several ways in which the Schrödinger model and Bohr model differ. Instead of allowing for continuous values for the angular momentum, energy, and orbit radius, Bohr assumed that only discrete values for these could occur (actually, quantizing any one of these would imply that the other two are also quantized).\)). It is interesting to compare the results obtained by solving the Schrödinger equation with Bohr’s model of the hydrogen atom. 
(b) The energy of the orbit becomes increasingly less negative with increasing n. 2 The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n. The absolute value of the energy difference is used, since frequencies and wavelengths are always positive. In this state the radius of the orbit is also infinite. U(r) ke2 r, where k 1 / 40 and r is the distance between the electron and the proton. The model has a special place in the history of physics because it introduced an early quantum theory, which brought about new developments in scientific thought and later culminated in. Why dont protons and electrons just slam into each other and explode Why do different elements emit light of different colors Niels Bohr knew why. In the electric field of the proton, the potential energy of the electron is. Historically, Bohr’s model of the hydrogen atom is the very first model of atomic structure that correctly explained the radiation spectra of atomic hydrogen. 
(b) Arrange the absorptions in terms of increasing energy. 9.4: The Bohr Model - Atoms with Orbits is shared under a CK-12 license and was authored, remixed, and/or curated by Marisa Alviar-Agnew & Henry Agnew. The Bohr model of the hydrogen atom ( Z 1) or a hydrogen-like ion ( Z > 1 ), where the negatively charged electron confined to an atomic shell encircles a small, positively charged atomic nucleus and where an electron jumps between orbits, is accompanied by an emitted or absorbed amount of electromagnetic energy ( h ). (a) Classify each transition as absorption or emission. With the assumption of a fixed proton, we focus on the motion of the electron. Some electron transitions are shown below on a Bohr-model representation of the H atom. 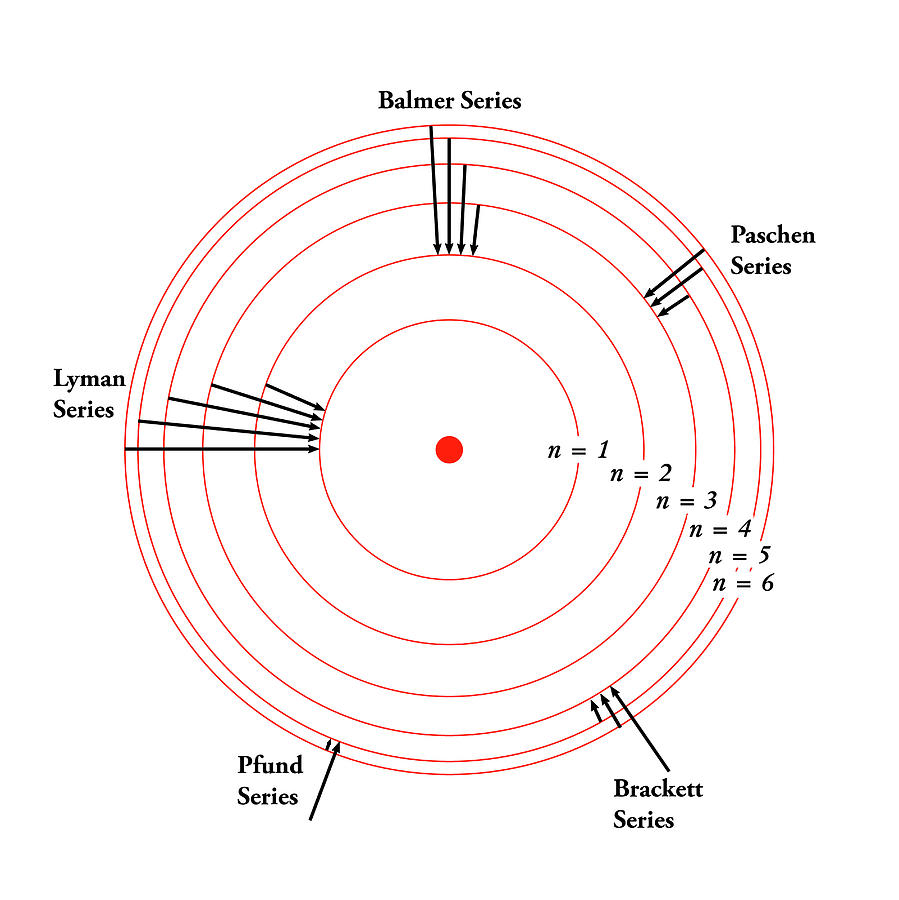
In this equation, h is Planck’s constant and E i and E f are the initial and final orbital energies, respectively. Figure 8.2.1: A representation of the Bohr model of the hydrogen atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
